Rate-based approach
There exist two basic stage models. The equilibrium stage model assumes that the flows leaving any column stage are in equilibrium. This assumption is combined with mass and energy balance equations to determine the concentrations and temperatures along the column. Another modeling approach for the description of separation units is the so-called rate-based approach. This method does not use the above-mentioned equilibrium assumption; instead, the process kinetics (mass transport, reaction, etc.) is directly considered in the balance equations. This approach has a more rigorous physical basis than the equilibrium model and a direct link to the column internals and design.
Therefore, we mainly work with the rate-based approach. The two-film model is applied for the description of the mass transport at the interface between two phases (e.g. gas and liquid phases as illustrated in the figure) [1]. All parameters of this model are largely available for different types of columns and internals in form of correlations. We have extended the two-film model for reacting and multi-component systems [2,3].
The simulation results have been validated against experimental data, which have been gained in several collaborative projects [4,5]. Non-reactive and reactive distillation columns (including dividing wall columns [6]) and absorption/desorption processes [7,8] can be successfully described by means of the developed models.
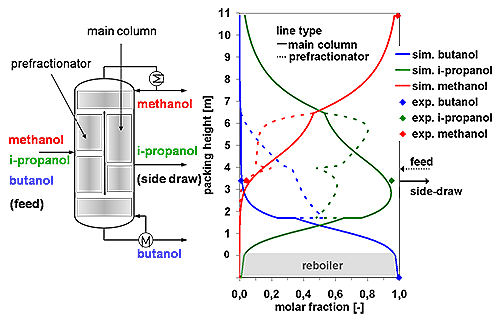
The following figure shows experimental data and simulation results for a dividing wall column separating a three-component mixture (methanol, isopropanol and butanol).
| [1] | Kenig, E.Y. Modeling of multicomponent mass transfer in separation of fluid mixtures Fortschr.-Ber. VDI, Reihe 3, Nr. 633. Düsseldorf: VDI-Verlag, 2000. |
| [2] | Kenig, E.Y., Kucka, L. and Górak, A. Rigorous modeling of reactive absorption processes. Chemical Engineering and Technology 26: 631-646, 2003. |
| [3] | Kenig, E.Y., Pyhälachiti, A, Jakobssen, K., Górak, A., Aittamaa, J. and Sundmacher, K. Advanced rate-based simulation tool for reactive distillation AIChE Journal 50: 322-342, 2004. |
| [4] | Kenig, E.Y. and Górak, A. Reactive Absorption In: Integrated Chemical Processes (Eds. K. Sundmacher, A. Kienle & A. Seidel-Morgenstern), Weinheim: Wiley-VCH, 2005. |
| [5] | Kenig, E.Y. and Górak, A. Modeling of Reactive Distillation In: Modeling of Process Intensification (Ed. F. Keil), Weinheim: Wiley-VCH, 2007. |
| [6] | Müller, I. and Kenig, E.Y. Reactive distillation in a dividing wall column: rate-based modelling and simulation. Industrial & Engineering Chemistry Research 46, 3709-3719, 2007. |
| [7] | Huepen, B. and Kenig, E.Y. Rigorose Modellierung und Simulation von Chemiesorptionsprozessen. Chemie Ingenieur Technik 77: 1792-1799, 2005. |
| [8] | Huepen, B. and Kenig, E.Y. Rigorous modelling of NOx absorption in tray and packed columns Chemical Engineering Science 60, 6462–6471, 2005. |